NeurOptimal finally admits not to be Neurofeedback
on 27 October 2018
Launched 7 months ago, the objective of my blog was twofold. On the one hand, I wanted to provide you with quality information on neurofeedback, but also and above all, I wanted to get information on the subject. It is often not discussed very much, but popularizers also learn a lot as a result of their work and it is this desire to learn that pushed me to get started.

On this existence, I can give you some statistics :
- the articles totaled more than 9,000 readings ;
- put end to end, they make 100 pages in A4 format ;
- the most popular article is "Finding, accessing, reading, reading, translating and understanding science", thanks in particular to the Zététique group where it was shared ;
- 150 emails were received via my contact form ;
- I met several of you to talk / introduce Neurofeedback and as a result, three of you followed the Neurosens Institute training (rather than the NeurOptimal one) ;
- I have been contacted by investment funds to discuss the scientific evidence of Neurofeedback and the Mensia Koala solution ;
- I won €0 through this blog.
Since March, a lot has changed in my approach to Neurofeedback and my understanding of the technique. My meeting with Ruben Perez and Thomas Feiner was very enriching and I am writing an article/interview of Thomas and his IFEN certification (an improved version of BCIA's) because I think it is important for the French people to know what is being done elsewhere in the world. I also secretly hope to be able to interview you Thomas F. Collura from Brainmaster, because in my opinion, that would also be very interesting.
But what has really changed is NeurOptimal. I had a lot of criticism and emails certifying that the NeurOptimal was Neurofeedback, that it was supported by scientific evidence, etc.... I spent time defending my evidence-based position and falling in the face of empty, argumentatively biased speeches.
Today, thanks to you (well, an anonymous source), I can finally tell you: NeurOptimal is changing its communication, recognizing the therapeutic effectiveness of Neurofeedback and recognizing that it is none of the above. Finally, according to a NeurOptimal representative and instructor.

In this e-mail, five elements are addressed :
- NeurOptimal would have been "evaluated" by the FDA ;
- Neurofeedback is a therapeutic practice ;
- NeurOptimal is a practice of "well-being" (who knows why the quotation marks are used) ;
- We must now call the technique underlying NeurOptimal as "Dynamical Neurofeedback" ;
- NeurOptimal is committed to stop using scientific studies about Neurofeedback to promote its own solution.
So, you know me now, let's go through it with a fine-tooth comb.
NeurOptimal evaluation by the FDA
The FDA is the Food and Drug Administration. According to Wikipedia (which we like, eh), "this organization has, among other things, the mandate to authorize the marketing of drugs in the United States".
And this is the first wall I've ever eaten myself. Why would an organization that manages drugs and food be interested in NeurOptimal? I mean, look at the FDA website. There are tabs for food, medicine, medical devices and lots of other very serious stuff.
When I read the FDA's English Wikipedia page (more detailed), I find nothing about a classification that might correspond to NeurOptimal.
But I'll be honest: it doesn't seem extraordinary to me that NeurOptimal has been "evaluated" by the FDA: this administration is perhaps larger than I think and we need to define precisely what this "evaluation" implies. It may just be paperwork to say that it is safe, without assuming any effectiveness. That is quite possible. So I searched the NeurOptimal website. Nothing. Nada. However, I noticed that they had removed their "Research" tab from the site (although the pages are still online using their search engine) and their communication has been changed.
I still played on the FDA website, entering "NeurOptimal", "Zengar" and "Neurofeedback" everywhere, but I invariably came across the message "no records were found with Device: neuroptimal Decision Date to 10/27/2018"... so here it is.
I still continued my investigation on the Internet, because so far, all the documents I have stipulated that NeurOptimal has not been evaluated by the FDA (for medical purposes).
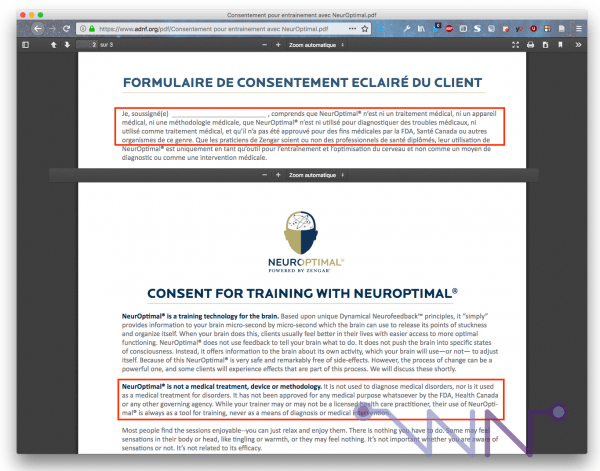
This type of consent form is still available on the Internet, including on the ADNF website, today :
However, by digging a little deeper, I found this document, which seems to be the only one that addresses FDA approval of NeurOptimal:
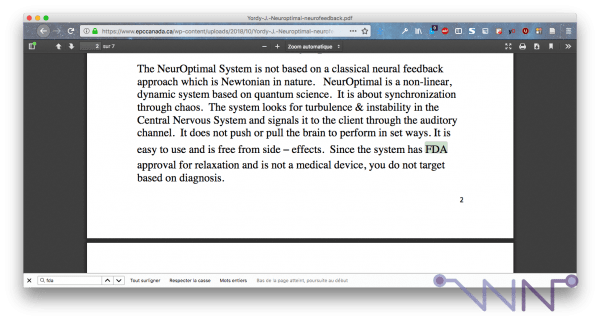
Here, it is mentioned that NeurOptimal has received FDA approval for use in relaxation. This is a little far from my previous articles, where I noted that NeurOptimal's communication advocated her ability to treat a whole bunch of pathologies. And I also pointed out in this article that NeurOptimal could be used as a relaxation device, as one asks the person to be calm. I like it when people say I'm right, a little thought for yesterday's me who worked well.
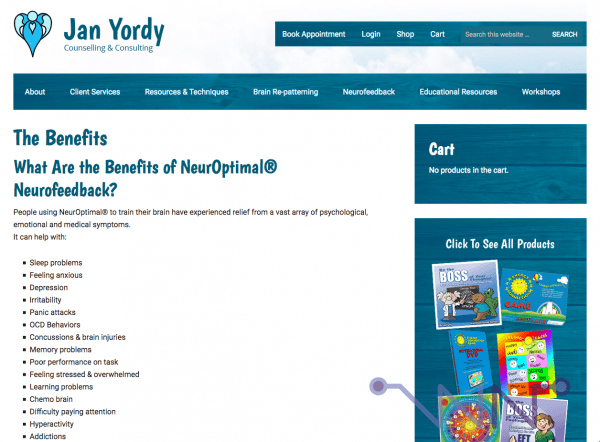
This document is the flyer of a lecture given by Jan Yordy, who holds a double master's degree in social work and education -she is neither a doctor nor a scientist- at the 20th Annual Canadian Conference on Energy Psychology. Is this something serious? If you want:
- follow healing touch sessions ;
- learn to diagnose and heal yourself through your dreams ;
- wake you up to your internal energy wiring ;
- train you in radiesthesia (capturing the "energies" that surround us, a big newage bullshit) ;
- communicate with your soul ;
- or..... talk about quantum DNA.....
Well, you've come to the right place. But this is no place for me. I am too young to commit suicide following a complete loss of faith in humanity in front of these conferences.
Then, on Yordy's website, she presents the NeurOptimal as a technique that will cure you of all ills, with of course, on the right, your cart, because Yordy, she is there to sell you things, above all.
So, it's not very serious.
But I said it earlier: honestly, I don't think NeurOptimal is lying about the FDA's evaluation of NeurOptimal. But in my opinion, it is oversold, so it is important to understand exactly what it is and how it works.
However, it must be understood that by using a search engine with the keywords "NeurOptimal" and "FDA", we invariably come across sites that talk about the fact that the FDA has recognized neurofeedback effective in stress management. The big problem is that all these sites share the same copied/pasted sentence (after all, they already do the same with their "user testimonials".......) :
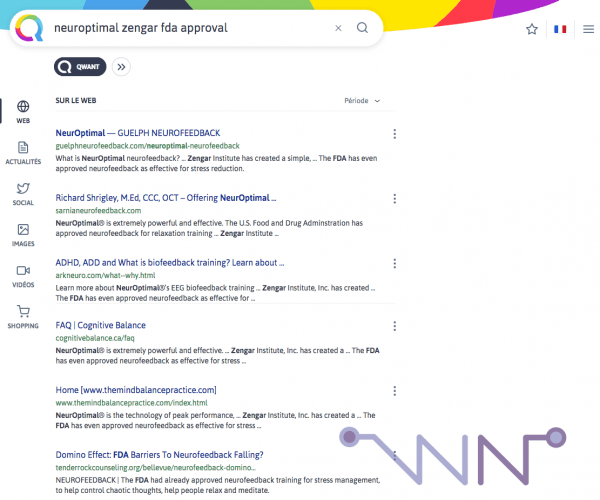
This gives in full version, once you go to these sites :
The U.S. Food and Drug Adminstration has approved neurofeedback for relaxation training and the American Pediatric Academy has endorsed neurofeedback as a “Level 1/Best Support” treatment for children with ADHD. (...) Because neurofeedback has been time-tested, it is also very safe. Respected institution such as the U.S. FDA and the American Academy of Pediatrics have endorsed neurofeedback.
The problem is that, to my knowledge, the FDA has not approved Neurofeedback (on the other hand, Biofeedback is subject to regulation and the EEGq NeuroGuide database is approved as well as many EEG-(Neurofeedback) materials). It would be NeurOptimal that would be approved for relaxation. On the other hand, the American Pediatric Academy has clearly recognized that neurofeedback is a therapy of choice for ADHD. So, we have a sentence that happily mixes NeurOptimal and Neurofeedback and Zengar's representative tells us : it's not the same thing.
So I was a little confused at the time. But in the course of my research, I came across an FDA draft that talks about a new product category called "General Welfare: Low Risk Materials Policy -- Guidance for Industry and FDA Staff". It is a document dated mid-2016, which addresses guidelines for wellness products :
The CDRH defines general wellness products as products that satisfy the following two factors: (1) are intended solely for general wellness purposes, as defined in this manual, and (2) pose a low risk to the safety of users and other persons. General wellness products may include exercise equipment, audio recordings, video games, software, programs and other products that are generally, but not exclusively, available from retail establishments.
Further on, is specified :
"The first category of intended uses of general well-being concerns claims for the general maintenance or improvement of functions associated with a general health condition that make no reference to diseases. For the purposes of this guidance document, this first category of claims is :
- weight management,
- fitness, including products intended for recreational use,
- relaxation or stress management,
- mental acuity,
- self-esteem (e.g., devices with a cosmetic function that make claims related solely to self-esteem),
- sleep management, or
- sexual function."
BINGO! I had just found new evidence: an FDA document that talks about relaxation. So we have the definition of a product "General well-being", now the part on "low risk materials":
"The CDRH's general wellness policy applies only to low-risk general wellness products. If the answer to any of the following questions is YES, the product is not low risk and is not covered by this guidance.
- Is the product invasive?
- Is the product implanted?
- Does the product involve an intervention or technology that may pose a risk to the safety of users and others if specific regulatory controls are not applied, such as risks related to lasers or radiation exposure?
NeurOptimal is not invasive, nor implanted, and does not use any laser or radiation, so for me it fits this definition perfectly.
Therefore, NeurOptimal, according to the FDA, would be a "low-risk general well-being" product.
Except that, by searching on the Internet, for example on this site or on this one, you can read :
The FDA has finalized a directive stating that it does not plan to review low-risk "general wellness products", such as portable fitness monitors or sleep monitors, and will not require them to comply with regulatory requirements before and after marketing.
I continued to dig and came across this document, a presentation of 40 slides on this type of product, which seems to be along the same lines: there is no need for a statement to the FDA.
And let us not forget: as French people, the FDA's decisions do not concern us. Europe often has stricter policies than the USA. So it's a clue, a lead, not evidence. Because between the American approach and our European approach, there can be a world.
Here, I was lost. NeurOptimal certifies that the product is evaluated by the FDA and after my research, I find that this product category does not require an evaluation...
....
...
....
Well, so I asked the FDA directly, by sending them an e-mail.
As we saw in the article I did in collaboration with the AAPB, Americans don't laugh too much. I had to sign a paper committing myself legally with the AAPB and in the FDA email I received, is specified below:
This communication is intended for the exclusive use of the recipient(s) named in this correspondence. It may contain information that is protected, privileged, or confidential, and it should not be modified. It may not be disseminated, distributed, reproduced, or copied to persons not authorized to receive such information.
So I'm going to assume that I'm not allowed to show you a copy of the email I received.
What I can do, however, is to provide you with the text of my email and the email address to which I sent it, so that you can also do the same. I exchanged two letters with the FDA and got a response in both cases within 24 hours. They are more efficient than some French administrations.....
I have sent an e-mail to the Industry and Consumer Education Division whose e-mail address is on this page and sent this text :
Information about NeurOptimal device (by Zengar)
Hello,
I am looking for information about the NeurOptimal material (https://neuroptimal.com) of the company Zengar. It would have been evaluated by the FDA as a relaxation device.
Could you give me a link to check if the FDA has actually evaluated the NeurOptimal and what are your conclusions? In addition, is it an assessment of its effectiveness, or simply a statement of its safety?
Thank you,
Sincerely,
The answer I received is consistent with my research: I could not find any information about the NeurOptimal device in any of our public databases on medical devices, including the Registration and List database. So it's no surprise, NeurOptimal is not a medical device and the FDA only manages that.
The person suggests that I submit a Freedom of Information request, but since this request is not free, I will skip my turn.
What I suggest in my articles is that you look at the evidence and discuss it. I have brought some elements comparing NeurOptimal's statements with the reality of the FDA, it is up to you to make your own opinion, now.
But from my point of view, NeurOptimal is -at best- relaxation. Take meditation classes, a good book where you can get a massage in a spa. The price will be lower and you will be just as relaxed ;-)
Neurofeedback is a therapeutic practice (NeurOptimal is not)
This is the second big piece, NeurOptimal recognizes the therapeutic benefits of Neurofeedback. "Therapeutic" refers to a "part of medicine that deals with the means -medicinal, surgical or other- to cure or relieve diseases" (Larousse). So it's a word backed by medicine, based on science. If in France, Neurofeedback is not recognized as medicine (we do not treat, we do not establish a diagnosis and we do not practice medicine), no doubt that in view of the progress of scientific research, this practice will gain its letters of nobility and I hope that one day, diplomas / training will be imposed to practice Neurofeedback. If only to protect the patient, or even to allow the reimbursement -even partial- of training by Neurofeedback.
In contrast, NeurOptimal is presented as a practice of "well-being". When people talk to me about well-being, I think of a person lying down with two slices of cucumber on their face with someone massaging her. Or, to me in the sauna, to wonder how I can survive at 90°C in an ultra-poor water atmosphere. And to tell me that I would have to try whole body cryotherapy at -145°C (by the way, it's a real medical practice in the treatment of chronic pain, so relaxing), to see what it's like to live a 235°C delta temperature. Relaxation is a bit like that for me. You have a good time, you relax, you're more rested on the way out. But if you were ADD at the entrance, you are ADD at the exit. The same goes for epileptics.
So it's great, I'm going to stop receiving insulting emails telling myself that I'm only talking crap about the NeurOptimal. Finally, I touch wood and cross my fingers.
NeurOptimal is "Dynamical Neurofeedback"
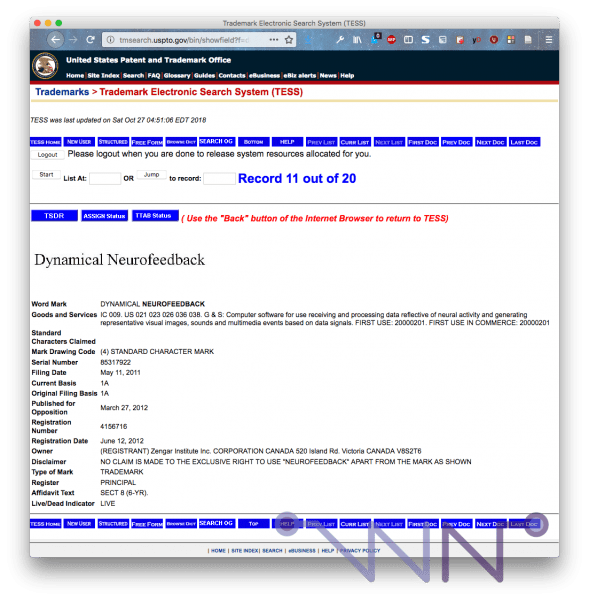
I have regained the trademark registration for the "Dynamical Neurofeedback". This is not new, it dates from 2011 and Zengar is kind because he specifies that he does not claim the exclusive use of the word "Neurofeedback". We didn't get far from the disaster!
So at the limit, I'd tell you, why not. Zengar is the only entity to speak of "dynamical neurofeedback" and "linear neurofeedback". He boasts of being the "first dynamical neurofeedback", but well, since it's the only one, it's easy. Crazy theories on the net, there are thousands of them.
In general, when I start talking to these people, I end up being insulted and called a scientist who is involved in a global plot where scientists would try to discredit these "innovative" theories. The person who doesn't speak English and who has never read a scientific article in his life (but he thinks he has relevant information for scientists, while he hasn't even read their writings), duh... and he wrote a book. As I was saying here, books are not proof of quality.
Again, all I'm suggesting to you, in these articles or when I'm talking to you, is to look at the evidence and discuss it. Proof is an experiment under controlled conditions, which can be analyzed objectively. That's not what anyone says, even if they're a scientist. It is an argument of authority, a rhetorical figure of manipulation.
In this example, between the theory largely denied by science (electromagnetism and DNA), the conspiracy approach (the dogmas of science are false), the absence of proof of what it claims, the big pharma conspiracy, or the non-evidence-based method of knowledge.... it was a winning third.
You know, I'm a nice guy, but I'm also direct and honest. I have no time to waste, no time to waste you. If you want to be credible: bring evidence. Read the scientific studies. Without it, you'll be full of shit no matter what, honestly.......
Despite everything, this time the person remained quite polite. I wish him to continue working, using science to refine what is currently an opinion, fed by fake news. Maybe not everything is good to throw away in the 200 pages of his book.
Even if this is an extreme case, well......
In the subject we are interested in, we are facing Zengar who sells a machine that is based on a concept not verified by science (because they do not communicate on it, easy, impossible to build proofs for or against) and who is convinced to save the world. You see the parallel with the previous case?
There is still a little something that bothers me about this notion of "dynamical neurofeedback". You know, it's like the French expression "homeopathic vaccine".
In one case, we have the vaccine, which is an extraordinary scientific breakthrough that has saved and continues to save millions of lives every year. On the other hand, we have a "homeopathic vaccine" that is only a sugar cube. There is therefore an extremely precise scientific term, with another invented word, that completely changes the meaning of the entire expression. And frankly, if it's not to mislead non-scientists, I don't know what it can be. For me, it is really intellectual dishonesty to use this kind of argumentative practice. NeurOptimal is not based on the principles of Neurofeedback, it is not Neurofeedback, why then insist on using this word in their communication if not to try to confuse people and try to benefit from the clinical effectiveness of Neurofeedback?
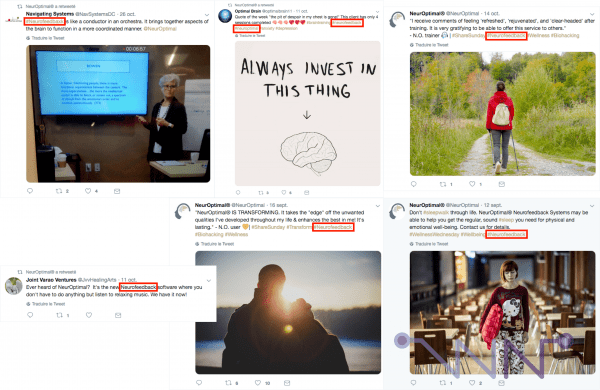
And besides, I went through NeurOptimal's communication a little bit and the confusion is still there today, skilfully maintained, as we can see with these very recent examples from NeurOptimal's Twitter page. In other words, hashtag #Neurofeedback is used extensively, even though it is no longer supposed to be.
Conclusion
One thing to keep in mind: beware of corporate marketing. Confront their allegations with reality. Words have a meaning, especially in marketing where it is their job. If you are told that a product has been "evaluated", ask yourself:
- was it the non-hazardousness that was assessed?
- is it the benefits provided?
- is it the scientific evidence?
- is it just the marketing speech?
In this case, I have the impression that it is simply the speech.......
During the writing of this article, I was contacted by a person, who was looking for advice, because she did not want to buy her "machine" at the Neurofeedback Training Institute where she had followed the training. It took me a few emails and a little insistence to get this person to admit that his "neurofeedback training" had been done at Zengar and that she was therefore absolutely not trained in Neurofeeback, but only in the use of NeurOptimal. She herself being convinced that she is really trained in Neurofeedback.
It's sad and I feel sorry for this person. As I said in a previous article, in general, people who practice NeurOptimal really want to help, they were just badly advised.
To conclude this article, I suppose, if I really wanted to be picky, I would publish a screenshot of the home page of the website of this person who writes in black and white "today we must absolutely dissociate Neurofeedback, which is a therapeutic practice, from dynamical Neurofeedback, which only concerns NeurOptimal®, a "well-being" practice".
Whoups....
Did you like this article? Then support the blog and share it with your friends by clicking on the buttons* below :
Article url :
http://en.chaussenot.net/neuroptimal-avoue-enfin-ne-pas-etre-du-neurofeedback
* These sharing buttons are respectful of your privacy and avoid tracking by social networks.




Avez-vous seulement essayé de faire quelques séances de Neurofeedback Dynamique pour vous faire une idée ???
Vous pourriez être surpris de ce que cela pourrait produire sur vous, bien au delà de la simple relaxation.
Parce qu'il y a des tonnes de preuves et d'expériences scientifiques sur certains médicaments par exemple, et pour autant ils sont délétères.
L'important n'est-il pas le résultat ???
Il est toujours possible de chercher à mieux prouver et expliquer... Toujours possible d'avoir le mot plus juste...
Votre débat n'est, en ce sens, pas inintéressant. Mais avant de critiquer et de dire que cela n'apporte aucun effet sur les TDA ou épileptiques, l'avez-vous seulement testé ???
C'est ce que j'ai fait, partant avec un a priori négatif, étant sceptique. Et les effets ont été inespérés. Le fait qu'ils soient inexplicables ne veut pas dire qu'il n'y a pas d'effets.
Je pourrais en dire tout autant sur la ritaline qui est prescrite à tort et à travers, qui a des résultats inexistants voir négatifs.
Pourquoi tant de hargne sur une technique qui a le mérite de faire enfin du bien et qui ne peut être cataloguée tellement elle est globale?
Honnêtement, il pourrait être intéressant de vous laisser surprendre par les effets en faisant quelques séances.
On ne parle bien que de ce que l'on connaît bien, non?
La question de mon témoignage concernant mon expérience avec NeurOptimal semblant importante pour vous, je vais y répondre : oui, j'ai essayé NeurOptimal.
A vrai dire, j'ai découvert NeurOptimal (il y a plus de 5 ans) dans une association -où je suis bénévole- qui a eu l'un des premiers systèmes en France.
Notre vice-président était un représentant et instructeur NeurOptimal, qui a d'ailleurs fondé un centre NeurOptimal. Suite aux discussions que j'ai eu avec lui et probablement d'autres choses, il a complètement lâché Zengar et NeurOptimal (pour partir sur une autre pratique probablement aussi peu scientifique, mais passons).
Alors, je me dis, si de mon expérience personnelle, je n'ai eu aucun effet de NeurOptimal et que si la personne qui a nous vendu le système (et faisait des formations dessus) il y a plus de 10 ans, lâche à présent NeurOptimal, qu'en penser ? Que ce système ne fonctionne pas. Voilà mon témoignage.
Mon côté scientifique vous parlerait de l’impossibilité du fonctionnement du système et des allégations mensongères, mais c'est le sujet de mes autres articles que je vous invite à lire.
Donc oui, je connais bien NeurOptimal, j'ai utilisé la machine et j'ai eu un représentant et instructeur pour en discuter avec moi. Je l'ai même utilisé pour voir comment elle fonctionnait.
Sur la dénégation de la science et de la médecine dont vous faites preuve, je ne vais pas réagir. Je ne comprends pas comment les gens arrivent, à ce point, à remettre en doute la méthode qui a permis l’émergence de la médecine moderne. Après, mon objectif est d'informer ceux qui doutent, pas de rallier ceux dont les croyances sont cristallisées et hermétiques à toute discussion basée sur les preuves.
Et jusqu'à présent, je n'ai aucune preuve de l'efficacité de NeurOptimal. Donc non, ce n'est pas une "technique qui a le mérite de faire enfin du bien et qui ne peut être cataloguée tellement elle est globale", car cela est une affirmation apportée sans preuve, qui peut donc être réfutée sans preuve. C'est la base de l'esprit critique, que j'essaie d'enseigner à mes lecteurs, certes sous le prisme de cet exemple précis.
Et sinon, pouvons-nous discuter du fait que vous n'êtes pas une personne contente de NeurOptimal (comme semble le faire croire votre commentaire), mais une personne qui vend des formations NeurOptimal et vend des séances de NeurOptimal (en usurpant les recherches sur le Neurofeedback sur votre site, ce que Zengar vous demande à présent d'arrêter) ?
Je n'aborderai pas votre formation de psychanalyste, mais sachez que j'ai longuement étudié cette pratique au cours de mes travaux de thèse et que la psychanalyse aussi, est une pratique pseudoscientifique. Qui a fait énormément de mal aux autistes en France et il me semble que la profession est au cœur d'une polémique actuellement, pour son refus d'appliquer des principes issus de la science.
Déclarer vos conflits d'intérêts à mes lecteurs est la moindre des politesses, plutôt que d'essayer de les manipuler avec un commentaire incomplet.
On ne parle effectivement que de ce que l'on connait bien et visiblement, vous connaissez mal la science et la médecine.
Cordialement
David Douçot
Je vous remercie de votre message qui comporte majoritairement :
- des insultes à mon égard ;
- un jugement de mes compétences (chose que je ne me permets pas vis à vis des praticiens de NeurOptimal) ;
- un jugement de mes attentes scientifiques (qui sont marquées clairement sur ce site et n'incluent pas du tout de reconnaissance, contrairement à ce que vous dites) ;
- encore une fois une pré-supposée efficacité du NeurOptimal basée sur les témoignages ;
- une dénégation de la communauté scientifique et une modestie quasi-inexistante ;
- un travestissement de la méthode scientifique que vous essayez de faire passer pour pseudo-scientifique.
En discutant avec des professionnels de la santé, on note aussi que votre message relève de la manipulation, avec des techniques classiques décrites dans les études de psychologie.
Cela se traduit par un déplacement du débat factuel sur le terrain émotionnel pour tenter de faire croire que le monde est contre moi, visant in fine à un apitoiement sur mon sort, mais comme vous avez bon cœur, vous me souhaitez malgré tout bonne continuation.
Ces techniques de manipulation ne fonctionnent pas ici et sont typiquement utilisées comme ligne de défense des manipulateurs qui se sentent menacés.
Alors, merci de vous exposer en public, cela servira également à mes lecteurs.
Au final, vous ne m'apportez aucune information sur NeurOptimal, son mode de fonctionnement ou même des études en conditions contrôlées qui me permettraient de revoir mon jugement. Voir sur le sujet de la FDA ou la confusion NO/Neurofeedback sur votre site (alors que vous demandez à votre communauté de ne pas le faire par mail), qui sont quand même centraux dans mon article... votre commentaire est donc vide de sens.
C'est pourtant tout ce que j'attends, car mon objectif est d'informer. Cet espace commentaire est là pour informer les lecteurs, donc vous pourriez très bien leur apporter des éléments factuels sur la méthode ? Des études ?
Donc, que mes utilisateurs voient réellement ce que les experts de NeurOptimal disent et veulent montrer de leur démarche.
Croyez-moi, si j’avais voulu vous insulter je n’aurais pas employé ce vocabulaire là. Donc non, je ne vous ai pas insulté, car votre avis ne m’interesse d’ailleurs pas du tout en réalité.
J’ai juste évoqué au passage une étroitesse de votre vision qui ne vous fait entrevoir les choses qu’à travers un prisme obsessionnel. Selon moi, vous devez être sacrément inoccupé pour vous permettre cela. Inoccupé et très mal renseigné sur le sujet, point. De plus, dans les grandes lignes, je n’ai aucune raison de vous contredire puisque vous dites vrai: NeurOptimal® est un outil de bien-être. C’est bel et bien ce que nous revendiquons, donc, dans un sens, vous vous battez tout seul contre vous-même ! Je vais vous donner un exemple très simple, personnellement, je préfère le vélo à la moto:
-1 les deux sont des moyens de transport qui nous amènent d’un même point A au même point B. (Le temps de transport n’étant pas un facteur crucial)
-2 la comparaison est vaine car tous deux ont des qualités et des points faibles, nous le savons tous.
-3 je ne prétends pas qu’un vélo soit une moto.
-4 je n’empeche personne de faire de la moto donc j’attends des motards qu’ils me foutent la paix quand je fais du vélo.
-5 je ne cherche pas à faire la course avec une moto, ce n’est pas mon but (qui reste principalement la balade).
Je veux par cet exemple un peu idiot de ma part, vous montrer à quel point vous vous battez contre des moulins à vent sans avoir pourtant suffisamment d’ouverture d’esprit, de spiritualité, d’imagination et de poésie pour le faire. Je ne vous en empêcherai pas, cela vous regarde.
Je ne prétends pas avoir plus de maturité que vous sur le sujet monsieur Chaussenot, je dis juste que notre point de vue et notre regard diffèrent et que cela ne fait pas de nous des ennemis qui devraient par définition s’opposer l’un à l’autre.
Je respecte votre pratique professionnelle et vos outils de travail même si je prétends qu’ils sont très largement aujourd’hui dépassés, mais vous remarquerez que je ne les attaque pas !
En retour, j’attends de vous la même chose, qui entre nous soit dit, ne peut que vous grandir davantage.
On ne sort jamais glorieusement vainqueur de s'être sali les mains à enfoncer quelqu’un dans la boue.
Cordialement
David Douçot
Je prends bonne note que mon avis ne vous intéresse pas, donc que vous passez ici du temps pour une chose qui vous semble inutile.
Je prends également bonne note que vous confirmez que le NeurOptimal n'est pas validé par la FDA, contrairement à ce que vous écrivez à votre communauté.
Je prends également bonne note que vous confirmez que NeurOptimal n'est pas du Neurofeedback, contrairement à ce que vous écrivez sur votre site.
Sur mes qualités propres, cela ne vous regarde pas, en effet, nous sommes d'accords.
Je note aussi qu'aucun vendeur de vélo n'a jamais fait la promotion de son produit en utilisant les caractéristiques d'une moto (vitesse, distance parcourue....), contrairement à ce que vous faites avec le NeurOptimal et le Neurofeedback, la métaphore n'est donc, absolument pas valide.
Partant des trois points d'accord que nous avons :
- le NeurOptimal n'est pas validé par la FDA ;
- le NeurOptimal n'est appuyé par aucune étude scientifique sérieuse ;
- le NeurOptimal n'est pas du Neurofeedback.
Je continuerai, via mon réseau, de mettre en lumière tous les praticiens NeurOptimal qui usent des études sur le Neurofeedback pour promouvoir la solution NeurOptimal.
Je continuerai également, de me renseigner sur l'aspect juridique concernant NeurOptimal, qui prône une efficacité sur des maladies (dépression, TDA par exemple, dans le cadre de votre site) alors que c'est du matériel non-médical, qu'aucune étude scientifique n'appuie ces allégations et voir si cela peut-être considéré comme de la publicité mensongère ou de la pratique illégale de la médecine.
Je respecte votre pratique professionnelle et votre outil de travail, en revanche, si celui-ci utilise les résultats du Neurofeedback pour se promouvoir, je serai présent pour le dénoncer.
NeurOptimal est un outil de bien-être qui n'a aucune action médicale démontrée et ne repose pas sur le paradigme du Neurofeedback, vous l'avez confirmé : à présent, que votre communauté se mette à jour pour refléter cela. Tant que ce ne sera pas fait, je continuerai à dénoncer vos pratiques, qui nuisent à la pratique du Neurofeedback.
votre entêtement, probablement commandé par votre hiérarchie (je me demandais à qui profitait le crime, j’ai eu ma réponse) comme j’ai pu le constater plus tôt dans la journée sur votre forum, vous fait inventer des choses que je n’ai jamais écrites, dites ou pensées !
-La FDA a validé NeurOptimal® comme outil de bien-être.
-NeurOptimal©️ appartient à la catégorie du «dynamical Neurofeedback®«
(La pratique du neurofeedback n’apartient pas qu’à la société pour laquelle vous travaillez)
Là où je vous donne raison, c’est que Neuroptimal©️ n’est pas une thérapie.
(Relisez bien ces quelques lignes, copiez les et diffusez les).
Quand je dis que vous confondez tout, en voici en exemple:
-Une bonne alimentation contribue à une meilleure santé: Cela ne fait pas d’une tomate un médicament ?
Nous sommes d’accord.
-le yoga contribue à être moins stressé: Cela ne fait pas du yoga une thérapie ?
Nous sommes d’accord.
-le sport améliore la santé générale: Cela ne fait pas du sport une médecine ?
Nous sommes d’accord.
-NeurOptimal©️ est une méthode d’entrainement cérébral qui contribue à tout cela: Ça n’en fait ni un médicament, ni une thérapie, ni une médecine ?
Nous sommes d’accord !!!
Il n’empêche que cela y contribue, que vous soyez d’accord ou non !
Cordialement
Alors, là, vous m'intéressez. Pouvez-vous m'apporter des preuves que la FDA a validé NeurOptimal comme outil de bien-être ? Je leur ai demandé directement et ils me répondent que non. C'est donc votre parole contre celle de la FDA. Si vous m'apportez la preuve du contraire, je dois modifier mon article qui en deviendrait faux.
Il me faut donc :
- une preuve de cette validation (un document, un lien sur le site de FDA, bref, quelque chose de vérifiable)
- les critères d'application de cette validation (est-ce l'efficacité qui est validé ? La non dangerosité ? Le discours marketing ?)
Mon formulaire de contact vous est ouvert, je vous remercie d'avance.
Je comprends bien le flou que vous voulez entretenir avec l'expression "neurofeedback dynamique", cela ne me dérange pas : ce qui me dérange, c'est quand sur un site NeurOptimal, vous utiliser allègrement le terme "neurofeedback" seul, ce qui pousse à la confusion.
Sur les exemples que vous donnez, l'impact de l'alimentation, du yoga et du sport ont fait l'objet de validation scientifique. C'est donc correct. Pour NeurOptimal, aucune validation scientifique n'existe. La métaphore est donc encore une fois fausse.
Donc, retirez toutes références à des maladies sur vos sites, ce n'est pas correct, cela ne se base sur aucune preuve, si ce n'est votre témoignage. Et un témoignage n'est pas une preuve. Et encore une fois, ce n'est pas à moi d'apporter la preuve de l'inefficacité de NeurOptimal, c'est à vous d'apporter la preuve de son efficacité, via des études sérieuses.
Et je le redis encore une fois : je suis sous le statut de micro-entrepreneur, mes seuls revenus viennent de ma pratique du Neurofeedback avec mes clients. Je n'ai pas de hiérarchie, je suis à mon compte. Et je n'ai aucun conflit d'intérêt avec des instituts de formation, des fabricants de matériel de Neurofeedback ou que sais-je.
Arrêtez de voir du complot partout, je suis simplement un praticien en Neurofeedback qui essaie d'éviter qu'une solution non basée sur les preuves scientifiques méprennent les gens.
Comme je vous le disais, les praticiens NeurOptimal utilisent les résultats scientifiques et la terminologie du Neurofeedback sur leurs sites. Dissocier proprement votre pratique de la mienne et effectivement, je n'aurai plus rien à dire.
Mais tant que ce ne sera pas le cas, votre publicité sera considéré comme mensongère et proche de la pratique illégale de la médecine, ce que je le redis, je dénoncerai afin d'éviter aux gens de confondre les deux.
je vous savais naïf mais là....
C’est drôle, je sors d’un entretien avec un haut dirigeant d’une association d´ethique Médicale en France:
NOUS, praticiens NeurOptimal©️, ne risquons absolument rien quant à la pratique illégale de la médecine, puisque nous sommes reconnus technique de bien-être (donc rien en commun avec la médecine) et le revendiquons haut et fort.
Par contre VOUS, c’est tout autre chose: vous n’etes PAS thérapeutes alors que vous prétendez l’être.
Oui je parle de naïveté car je ne sais pas qui vous a fait croire qu’une technique même sous ordonnance d’un médecin était une technique reconnue par la médecine... qu’une technique en etude dans les hotpitaux était reconnue par la médecine, qu’une technique dont certes les études sont nombreuses était une technique reconnue par la médecine !!! TOUT ÇA est faux monsieur Chaussenot.
C’est peut-être vrai ailleurs mais en France c’est faux.
Seule une pratique dite thérapeutique mais encadrée par un médecin lui même présent dans cette pratique est autorisée afin d’être étudiée et reconnue.
Donc je vais me répéter:
nous ne formons pas des thérapeutes et sommes une technique de bien-être donc ne craignons rien puisque nous ne prétendons PAS appartenir à la catégorie thérapeutique contrairement à vous qui vous exposez en France précisément à ce genre de danger. Ça paraît pourtant simple à comprendre mais visiblement pas pour tout le monde.
Dernier détail renseignez-vous un peu mieux quant à notre appartenance à la FDA.
(Je vous imagine repeser méticuleusement votre KG de tomates une fois arrivé à la maison en prétextant que le vendeur n’a mis que 998g en criant au scandale et en l’insultant d’être un voleur).
C’est insupportable !
Bref quand vous l’aurez trouvée cette preuve tant attendue (par vous seulement), faites en part à vos quelques lecteurs.
Enfin sachez que vous pourrez me répondre tant que vous voudrez, me donner tort, nous donner tort, nous traiter de tous les noms... je n’ouvrirai plus votre blog qui m’a amusé ce jour mais qui m’a également fait tellement perdre mon temps.
Donc en gros, dès que je demande des preuves, c'est le moment de fuir.
Même quand la FDA dit l'inverse de vos allégations.
Bon, sujet clos donc, aux lecteurs de se faire leur avis.
Merci pour l'échange :-)
Sorry because my intervention is not in French, but I feel more confortable in English. Well, as far I know, testimonials are not a scientific proof. Are scientific studies an absolute truth? Absolutely NO. But they are better than just opinions. I don´t know what Dynamic Neurofeedback is, or what non-linear neurofeedback is...and I don´t know what they are because (right now) I have written the both terms in PubMed and I didnt find anything. The use of leeches in the Middle Ages also had very good opinions but it was not a relief for any disorder. I do not want to make a criticism but in science we like to try things with facts not opinions. I hope no one is offended, and that the search for facts is intensified using the methods that we can all replicate
Bonsoir,
Désolé parce que mon intervention n'est pas en français, mais je me sens plus à l'aise en anglais. Pour autant que je sache, les témoignages ne sont pas une preuve scientifique. Les études scientifiques sont-elles une vérité absolue ? Absolument NON. Mais elles sont meilleures que de simples opinions. Je ne sais pas ce qu'est le Neurofeedback Dynamique, ou ce qu'est le Neurofeedback non linéaire... et je ne sais ce qu'ils sont parce que (pour l'instant) j'ai écrit les deux mots dans PubMed et je n'ai rien trouvé. L'utilisation des sangsues au Moyen Âge avait également de très bonnes opinions, mais ce n'était un soulagement pour aucune maladie. Je ne veux pas faire de critique, mais en science, nous aimons éprouver les choses avec des faits et non des opinions. J'espère que personne n'est offensé et que la recherche des faits est intensifiée en utilisant les méthodes que nous pouvons tous reproduire.
Je suis américaine et une maman de 3 enfants.Nous vivons en France depuis 3 ans. Mon fils de 14 ans est autistic. Je dois dire que je suis juste scandalisée de lire votre blog! Pourquoi voulez vous détruire un appareil qui apporte de l'espoir la ou justement la médecine ne peut rien faire? Je pense moi au contraire que ce sont les résultats qui comptent, les chiffres restent des chiffres qui bien trop souvent sont déformés parce que les études sont financées par les concepteurs eux mêmes. Lorsque vous vous retrouvez dans des situations sans espoir et que vous ne voulez pas baisser les bras des appareils comme le neuro optimal apportent une solution. Il y a deux ans alors que j'étais bien depressed j'ai trouvé ce système et aujourd'hui grace à cela mon fils communique avec nous. Il était complètement en souffrance, ne communiquait pas du tout et avait régulièrement des effondrements. Il pleurait beaucoup pour rien. Cela a affecté toute la famille. J'étais en depression profonde, croyez moi avoir un enfant différent c'est très dur. Aujourd'hui même si bien évidement l'autism est toujours la, nous arrivons à communiquer avec notre fils, il joue, dessine et a fait d'énormes progrès. Je suis sortie de la dépression et je le dois complètement à cette machine. Qu 'elle soit médicale ou non ne m'intéresse personnellement pas. Ce qui compte c'est le résultat . Alors de lire votre jargon de médecin me rend malade. N'êtes vous pas sensé aider l'humanité? Ou cherchez vous à descendre cet appareil parce que vous en vendez vous même? J'espère que ce commentaire vous arrivera car je ne doute pas que vous l'effacerez bien vite! Je vous souhaite bonne chance dans votre combat...avec vous même.
MB
Votre témoignage ne sera pas effacé, chacun est libre de s'exprimer.
En revanche, je possède des millions de témoignages sur le fait que l'homéopathie soigne des maladies, certains disent même que ça a soigné leur cancer. Dois-je proposer de l'homéopathie à mes amis qui ont le cancer ? Non, car la science a démontré que c'était similaire à un placebo, je risquerai de leur faire plus de mal que de bien. Il est fort probable que le cancer ait été soigné avec autre chose.
Dans le cas NeurOptimal, c'est pareil. Il existe d'autres solutions pour traiter le TDA et la dépression, des solutions qui ont fait leurs preuves au niveau scientifique, avec des financements publics et sans aucun conflit d'intérêt. C'est ce vers quoi je vais diriger les gens avant tout.
Si un jour NeurOptimal fait ses preuves au niveau scientifique, mon discours changera. Mais à l'heure actuelle, je ne vois aucune bonne raison de conseiller un appareil qui n'a pas fait ses preuves alors que d'autres approches ont elles, fait leurs preuves.
R.
Mais tout de même, que proposez vous aux gens qui n'ont pas d'autres solutions ou qui n'ont plus rien à perdre?
Je ne pratique pas l'homeopathy mais je connais quelqu'un qui soigne sa thyroid grâce à cela depuis 10 ans car elle ne supporte aucun autres médicaments. La science a peut être démontré que c'était similaire à un placebo mais en attendant elle vit bien et ces millions de gens qui prennent de l'homeopathie aussi apparement! Quant aux placebos cela ne s'applique pas à TOUS les médicaments peut être? Les études faites à ce sujet auraient elles tort?
Et puis je doute fort qu'un enfant autistic réagisse par effet placebo au neuro optimal. Savez vous qu'ils sont des centaines d'enfants comme mon fils à avoir eu des effets inespérés grâce à cette machine? Je trouve criminel de vouloir salir une telle invention. Je ne comprends pas en quoi cela vous dérange? ...a moins que VOUS ayez un conflict d'interrêt !
MB
Votre discours ressemble beaucoup aux autres commentaires des partisans de NeurOptimal que j'ai eu sur ce blog. Mon rôle ici est d'apporter des preuves factuelles et concrètes. Vous, vous n'abordez que des témoignages invérifiables en campant sur vos croyances.
Cette discussion n'apporte donc aucun élément intéressant à mes lecteurs.
Je ne vois rien de plus à ajouter que ce que j'ai abordé dans mes articles et mes commentaires précédents qui répondront à tout ce que vous abordez dans votre commentaire.
R.
J ai fait pratiquer du neurooptimal sur mon enfant. Cela lui a fait du bien.. Mais j attendais plus, de résultat sur ses difficultés réelles.. Quand j ai décidé d en faire on m a informé que C était du neurofeedback !! Et en faisant des recherches sur internet.. Il y a confusion.. Donc j ai eu le sentiment de m être fait avoir.. C est pas clair.. Si je l avais su dès le départ.. Je n aurai pas autant dépenser.. Et je ne l aurai pas fait.. Je ne critique pas le neurooptimal mais je le reproche d utiliser le terme de neurofeedback.. Continuez de nous informer..
Cordialement
Si je comprends bien, vous êtes praticien en neurofeedback et non en neurofeedback dynamique de neuroptimal (Zengar). J'aimerai donc savoir quelle technique, appareil, méthode vous utilisez , quel est votre protocole de soin pour les enfants tdah, quels sont vos résultats (nombre de séances et régularité des séances) ainsi que le tarif que vous appliquez. Ma fille de 7 ans et demi est HQI 136 avec tdah important, elle a donc de grosses difficultés dans ses apprentissages scolaires car elle n'arrive pas à se concentrer, s'énerve très vite et gère difficilement ses émotions. Je suis attentive à ce que vous écrivez car évidemment, je cherche une solution pour aider mon enfant à se sentir mieux dans sa peau (impact énorme sur la famille) et j'en ai marre de dépenser une fortune à droite à gauche dans le vide. Ma fille a eu une 1ère séance avec neuroptimal (je cherchais du neurofeedback sans connaître les différences qui existent) à 35 euros la séance et la 10ième offerte et cela, à 5 mn de chez nous en province. On n'a pas beaucoup de choix par ici. Bref, selon vous, si vous étiez parent d'un enfant tdah qui souffre depuis des années et qui est déjà capable de vous parler de suicide à même pas 8 ans, si vous étiez ce parent démuni, qui se bat pour trouver une solution sans ritaline, ce parent qui s'épuise nerveusement et physiquement, qui met sa carrière en péril à cause de cet épuisement, qui heureusement, mais ce n'est pas le cas de tous, n'a pas divorcé malgré les difficultés quotidiennes amplifiées lorsqu'on a un ou des enfants en difficultés... que feriez-vous ? Je suis tentée par Mensa KOALA puisqu'il faut une ordonnance d'un médecin et que c'est spécifiquement pour le TDAH et que le système est reconnu comme médical (tarif d'environ 1800 euros). Il y a également l'option d'une autre praticienne à 20 mn de chez nous qui utilise l'EEGQ de Neurosens apparemment à 50 euros la séance soit pour 40 séances 2250 euros. Votre cv indique que vous travaillez pour une association mais celle-ci semble ne pas fonctionner, qu'en est-il ? Merci d'avance
Vous comprenez bien, effectivement, pas de NeurOptimal ici, qui n'est qu'un placebo de luxe.
A titre personnel, je ne suis pas ce parent démuni, je serai plutôt cet enfant, étant moi-même handicapé et ayant probablement vécu le même genre de scénario que votre fille. Je n'ai pas de solution miracle, à l'heure actuelle pour le TDAH. Tout comme il n'y en a pas pour ma pathologie.
Bien que ce blog vise à tenter de rassembler les gens qui sont dans une situation similaire, je n'ai pour le moment aucun retour sur Koala, donc je ne peux rien en dire. Ils ont du marketing, ils ont une belle étiquette, mais niveau preuve scientifique, c'est pour le moment inexistant.
Si la ritaline n'es pas une option pour vous, vous n'avez guère de choix :
- donner une chance à Koala, en se rappelant qu'actuellement il est testé pour un cas particulier : TDAH sans comorbidité ni médication. Vu que refusez la médication, c'est déjà un point positif.
- sur les élèves de Neurosens, cela va réellement dépendre du praticien. Le Neurofeedback basé sur l'établissement d'un profil cérébral + un entrainement spécifique (dans le cadre du TDAH, on possède deux protocoles en fonction du profil cérébrale de la personne) peut fonctionner. Après, j'avouerai ne pas connaître la personne qui exerce dans votre région, qui semble certes faire du neurofeedback, mais aussi d'autres techniques qui me semble scientifiquement discutable...
- sur l'AFBN, je l'ai quitté par manque de temps, ayant déménagé au Japon, donc cela ne regarde plus que les bénévoles actuels qui visiblement, en effet, n'ont pas beaucoup avancés cette année.
N'hésitez pas à me contacter par e-mail (formulaire de contact) pour avoir plus d'informations (voir s'appeler pour en discuter de vive voix), en gardant en tête, qu'étant actuellement à l'étranger, j'ai suspendue mon activité de NFB.
Rémi
Pouvez-vous, savez-vous quelle est le niveau de formation d'un praticien exigé par Neurosens pour qu'il puisse pratiquer l'EEGQ ? En d'autres termes, a-t-il obligatoirement une formation complète ou peut-il utiliser cette méthode dés lors qu'il a suivi une MINI formation et qu'il a les moyens suffisants d'investir dans du matériel ? Les mises à jour du logiciel, la qualité des électrodes et cie sont-elles obligatoires ? J'imagine qu'étant payantes les prestations de SAV ne sont pas toujours demandées par le praticien, tout comme les formations qui s'ajoutent à la formation initiale... (j'ai pu voir les prix du Neuroptimal d'une part qui représente un investissement financier important tout comme les formations et surtout les tarifs de suivi post vente en cas de soucis de matériel par ZENGAR sur le site de l'association française, et c'est exhorbitant)
Ce qui est inquiétant, c'est lorsqu'il s'agit d'un programme ouvert, le praticien se doit d'être réèllement engagé pour être compétent contrairement à un système fermé comme Neuroptimal ou Mensa Koala.
Mensa Koala ne précise pas le type de comorbidités qu'il exclu pour l'usage de son système et je trouve cela imprécis justement car la plupart des gens ont des troubles associés comme des TOC, une dépression au moins latente, ou autres...
Je me demande également si les normes de mesures électriques des neurones sont différentes chez une personne à Haut potentiel (surdouée) sachant que d'un point de vue neurologique on parle de neuro droitier (hémisphère droit + sollicité) pour les HQI et de neuro gaucher (hémisphère gauche prioritaire) pour les personnes non HQI. Dans ce cas, si différence de mesures électriques, le neurofeedback est-il non indiqué pour les HQI.... ?.....
Merci de me proposer une discussion de vive voix, je vous contacterai probablement par mail pour cela.
Cependant, notre échange ici peut profiter à tous vos lecteurs et je ne dois pas être la seule à m'interroger sur ces points.
Merci d'avance.
Côté software, des entreprises proposent des solutions abouties actuellement. Et les mises sont certes payantes, mais je l'espère, budgetées par les praticiens. Ce n'est pas un point qui m'inquièterait outre mesure.
De même, les électrodes, à partir du moment où on achète du matériel de qualité vont durer dans le temps. Et ensuite, si vraiment il faut les changes, c'est l'affaire de quelques euros.
En NFB, le plus coûteux, c'est l'équipement initial, la maintenance est vraiment ridicule à côté, à moins d'aller casser l'encodeur (qui lui coûte cher) ou l'ordinateur.
On n'est pas du tout dans le système pyramidal de NeurOptimal, mais dans un vrai commerce sérieux de matériel utilisé par des laboratoires scientifiques, des hôpitaux et des professionnels indépendants.
Sur Koala, ils excluent TOUTES les comorbidités. Des informations que j'ai, c'est ce qui explique que cela fait 2 ans qu'on attend leur étude clinique, car trouver de tels profils est rare. Mais il faut bien commencer par un cas simple, pour ensuite faire des études plus complètes en ajoutant les comorbidités / médication. Donc sur le plan clinique, cela peut-être choquant, sur le plan scientifique, c'est la bonne façon de faire. Comme je disais, le produit a été lancé trop tôt à mon goût (avant validation scientifique), principalement car il faut de l'argent pour financer le personnel / montrer patte blanche face aux investisseurs.
Sur le fonctionnement cérébral, on rentre réellement dans le coeur du métier de l'étude des cartes cérébrales et l'utilisation éventuelle de bases de données normatives (qui ne sont pas nécessaire normées sur le HQI, du coup).
Après, cette idée de neuro-machin, honnêtement, c'est complètement faux d'un point de vue des neurosciences et une idée qui a la peau dure.
A partir de l'établissement de la carte cérébrale, de sa comparaison à une population "normale" et des différentes études réalisées sur la pathologie cible, c'est là le boulot du praticien d'arriver à un protocole éprouvé, qui sera éventuellement modulé en fonction de la progression individuelle de la personne.
R.
Le LORETA est une technique solide et précise pour le NFB. Après, c'est une grosse formation, qui a un coût non négligeable et qui demande du temps. Donc c'est plus pour quelqu'un qui désire s'y mettre à temps plein.
N'hésitez pas à me poser vos questions par mail, ce sera peut être plus simple d'échanger par ce biais.
R.
Tout d'abord, merci et bravo pour la clarté de votre blog et de la rigueur de vos démarches. On reconnait l'esprit scientifique qui contraste bien avec certains commentaires à caractère viscéral.
Je suis en fin de doctorat dans les sciences des matériaux, et j'ai petit à petit développé le long de mes études en physique, un attrait paradoxal par tout type de domaine sortant soit de l'entendement, soit de la compréhension simple. Par contre, je place mon esprit critique toujours en alerte maximal afin de ne pas me faire happer par les moyens d'enfumages et les techniques marketing de personnes en conflit d'intérêt, flagrant ou dissimulé. J'ai eu l'occasion de côtoyer le monde du Neurofeedback (à remplacer par le terme qui convient ^^ ) par une intéressante praticienne qu'on a sollicité pour ma fille. Je vous fait pas l'affront d'amener ma propre expérience personnelle pour dire si oui ou non les séances ont changé ma fille ou non (par échange mail privé, pourquoi pas). C'est juste pour donner de la crédibilité sur mon expérience. J'ai également réalisé une séance personnellement et je cherche à approfondir le sujet afin d'en refaire ou non.
Je suis très satisfait de pouvoir lire (enfin) un avis dissident sur le sujet, car une telle technique décrite de cette manière sur internet, cela me donne l'impression d'y avoir que des avantages (sauf financier...), que des bienfaits, et zéro contre-indication. C'est précisément de là où vient ma réticence sur ce sujet et c'est la raison pour laquelle je suis très heureux de pouvoir vous lire et pourquoi pas échanger avec vous.
Bon, j'arrête de tergiverser, voici mes questions/remarques :
- Je n'arrive malheureusement pas à conclure, après avoir lu votre article, si les avancés scientifiques dans les neurosciences sont dans le sens d'une optimisation du cerveau et de ses connexions neuronales. Nous comprenons que vous tentiez de nous mettre en garde contre l'usine-à-gaz "Neuroptimal" mais sur l'apport de la technique sur des individus, est elle étudiée dans la littérature?
- Je suis surpris par un de vos commentaires, le premier, où suite à votre travail de recherche de vérité bien structurée, vous "semblez" utiliser votre expérience non concluante pour établir que ce système ne fonctionne pas (l'argumentation impliquait également le formateur ayant quitté Zengar). Que vous concluiez de là, qu'il ne soit pas global, j'entends, et c'est même mathématiquement correct de dire que grâce à un unique contre exemple, l'on casse une hypothèse contre 20 millions d'exemples. Mais vous en conviendrez que ce système axiomatique est assez loin de ce que nous vivons dans ce monde réel, avec toutes les approximations que cela implique. Non?
Que je sois dans le groupe des personnes pour qui cela a marché ou non, ne devrait pas influer le débat.
- La praticienne avec qui on a travaillé et sur qui j'ai assez confiance en sa bonne foi, m'a parlé également des "rythmes" ou des "modes" de fonctionnement du cerveau de manière général, qui pouvait changer d'une ondulation à une autre permettant d'être soit dans un stade de concentration, soit en mode travail machinal répétitif, etc... D'autant plus qu'elle m'expliquait qu'avec l'habitude, elle ne ressentait plus son propre cerveau comme une masse informe et inerte. Les sensations, pressions, et ressentis physiques (un peu comme si son cerveau appuyait sur telle ou telle zone du crane) pouvaient être interprétés ce qui lui permet même de pouvoir switcher d'un mode à l'autre ! ... (promis, si elle m'avait sortit le mot "quantique" à ce stade, je serais parti en gueulant !). Cela m'a paru étrange et douteux, mais pas impossible.
Avez-vous entendu parlé de ça également, et si oui, pourriez-vous m'en dire plus ou me diriger vers des articles/références?
Désolé d'avoir été aussi long.
Merci pour votre patience et votre temps.
Et encore bravo pour votre travail.
Bien à vous
Ron
- Sur le Neuroptimal, il est assez facile de savoir si cela a été étudié dans la littérature : une rapide recherche dans Pubmed sort 0 résultat sur "Neuroptimal", "Zengar" ou "Dynamical Neurofeedback". Mon interprétation de ce résultat est donc que non, le NeurOptimal n'a jamais fait l'objet de recherche de qualité. Ce qui fut confirmé dans mon article suivant par le fondateur de Zengar et créateur du NeurOptimal (https://chaussenot.net/neurofeedback-decrypte-formation-pratique-collaboration-ifen, en début d'article).
A partir de là, la technique ne doit pas faire d'allégations relatives à la médecine ou utilisant des noms de pathologies, car aucune étude n'existe sur le sujet.
- Sur mon commentaire relatif à mon expérience à Zengar, il est très probable que son seul objectif fut de me mettre au niveau des personnes défendant bec et ongle NeurOptimal (ie, utiliser un témoignage face à un témoignage, vu que la science ne se prononce pas sur le sujet). L'objet n'est donc pas tant de dire "NeurOptimal n'a pas fonctionné sur moi, donc il ne fonctionne pas" (ce qui est faux), mais d'expliquer que venir brandir un témoignage positif ne sert strictement à rien, car ben... en face, on peut brandir une expérience négative.
Mon opinion sur le NeurOptimal, c'est qu'il n'existe aucune étude à son sujet et donc aucune preuve d'une efficacité supposée. De plus, NeurOptimal ne repose pas sur des paradigmes scientifiques (le neurofeedback) et ne fait qu'usurper un titre "-Neurofeedback- dynamique" qui, à mon sens, ne sert qu'à duper le grand public, qui en faisant ses recherches tombera sur les résultats du Neurofeedback (non dynamique) et donc, pensera qu'effectivement, c'est sérieux.
De plus, partant de l'existence prouvée et pas mal étudiée de l'effet placebo (et contextuels) pouvant tout aussi bien expliquer les résultats du Neuroptimal (ou ceux de l'homéopathie), j'en déduis, me concernant que NeurOptimal n'est qu'un placebo onéreux, contrairement au Neurofeedback qui a fait ses preuves en double aveugle face au placebo.
- Sur le témoignage de la praticienne, ben, que dire ? C'est un témoignage. Certains entendent des voix de l'au delà (medium), d'autres ressentent les énergies (médecine chinoise, coupeur de feu), et j'en passe.... quelles preuves avons-nous de l'existence de ces sensations / ressentis ? A mon avis, aucune. Et là encore, l'auto-suggestion ou les mouvements involontaires conduisent au même résultat sans utiliser d'explication extraordinaire (ce qui a été étudié dans le cas des sourciers, par exemple). Donc, je préfère avoir comme opinion que rien ne me prouve l'existence des phénomènes rapportés par témoignage, indépendamment de la bonne foi de la personne (qui peut se tromper, même involontairement). Donc, les gens peuvent bien croire ce qu'ils veulent, mais pour me convaincre, il me faudra plus qu'un témoignage. Et ce n'est pas à moi d'en apporter la preuve, la charge de la preuve revient à celui qui effectue les affirmations.
Je vois beaucoup d'explications permettant à un individu de prendre conscience de son activité cérébrale ou d'en moduler le fonctionnement (par méditation, auto-hypnose, neurofeedback...) qui ont fait preuve d'efficacité, donc, je préfère penser en premier à cela, plutôt que d'une action du Neuroptimal.
De mon opinion personnelle, l'espèce humaine à un besoin maladif de sens et d'explications. C'est pourquoi, elle comble les trous avec ce qu'elle peut. De là, l'expansion des religions et des pseudosciences qui ne font que se répandre au dépend de la science : les gens sont plus intéressés par une affirmation extraordinaire qui donne du sens que par un fait scientifique qui explique pourquoi il ne faut pas jeter d'eau chaude sur un pare-brise gelé. Et une fois que l'on est convaincu d'une chose et il très difficile de faire machine arrière. Dé-croire ou ne plus croire demande beaucoup plus de preuves que les allégations initiales qui ont forgées cette croyance.
Donc ben, NeurOptimal, il y a des éléments divers, que j'essaie d'exposer ici, ensuite à chacun de se forger son opinion. La mienne me fait dire qu'aucune preuve scientifique ne prouve l'existence d'efficacité de la machine, donc à titre personnel, je préfère ne pas croire à son efficacité, jusqu'à preuve du contraire.
Merci à toi en tout cas !
R.
Un rapide commentaire pour vous dire un grand merci pour cet article, j'ai un blog et je suis actuellement entrain d'écrire un article qui parle des thérapies "neuroplastiques". Même si le neurofeedback est un peu à part, je souhaitais intégrer un paragraphe dessus mais j'avais peur que mes lecteurs contactent un thérapeute peu fiable. Les infos de votre article me donnent des bases solides pour valider mon pressentiment sur les systèmes Neuroptimal et co.
Savez vous à tout hasard, s'il existe une liste, un référencement quelquepart des hopitaux et thérapeutes privés utilisant le neurofeedback avec EEG et EEGq? Je comptais contacter Neurosens pour leur poser la question mais si vous avez la réponse je suis preneuse :-)
D'ailleurs puis-je intégrer un lien sur votre site dans l'article pour que les gens puissent avoir des infos supplémentaires?
Encore merci pour la clarté de vos informations
Aurore
Tout d'abord, un grand merci pour le partage de vos recherches, avis, mises en garde...
Je suis le "bon client" ignorant qui se ferait facilement avoir. Du fait de mon incompétence certes, mais aussi parce que je suis convaincu à 100% que le neurofeedback fonctionne (hum... incompétent et convaincu, pas très scientifique tout ça).
Je suis le papa d'un enfant TDAH + dyslexie + dyspraxie. Comme Bindia ci-dessus, je suis attiré par Koala (étant tombé sur un intervenant "TedX" Louis Mayaud sur youtube).
Mes questions :
-Vous parlez de comorbidité. La dyslexie et la dyspraxie sont-elles des comorbidités ? (j'ai "googelé" mais je n'ai pas trouvé la réponse).
-Le fait de faire le "traitement" tout seul à la maison (sauf pour la première séance si j'ai bien compris leur approche) vous semble-t-il éliminatoire ?
-Enfin et même si je pense que vous serez réticent à répondre : pourriez-vous me dire quel(s) produit(s)/software /méthodes/ vous recommanderiez pour aider un enfant comme le mien. Si vous êtes trop réticent, je reformule : quel(s) méthodes/produits/software/équipements/ ont à votre connaissance, fait la preuve de leur efficacité, ou à défaut d’efficacité prouvée, présentent un minimum de gage de sérieux du fait l'approche scientifique employée( et pas seulement du commercial et du marketing).
PS: je rejette la voie médicamenteuse... enfin...pour le moment...
Merci,
Cordialement,
Patrick
Actuellement, il n'y a, à ma connaissance, aucune liste fiable référençant les praticiens en Neurofeedback. Le problème sous-jacent, c'est qu'il n'y a aucun organisme fiable et indépendant qui peut maintenir une telle liste.
Neurosens vous redirigera vers sa propre liste, de praticiens qu'ils ont formés, mais l'hétérogénéité des compétences est à prendre en compte. J'ai des témoignages de patients passés par les mains de ces praticiens et certains sont négatifs, même si toujours bienveillants.
Aucun soucis pour intégrer un lien.
Merci à vous
R.
Concernant Koala, mon site n'est absolument pas à jour.
Des parents m'ont informé que Mensia a été racheté (de mémoire par la boite mère de Melomind) et qu'il est à présent très compliqué de se procurer Koala. Certains ont eu des soucis pour la facturation lors du changement de propriétaire. D'autres ont eu (après 2 ans !) une prise en charge MDPH.... mais Mensia n'existant plus, ils étaient laissés sans rien.
Du coup, même si Koala est une option, gardez en tête que si les problèmes de changement de propriétaire ne sont pas réglés, cela sera compliqué. L. Mayaud (ex-responsable scientifique Mensia) a d'ailleurs quitté le navire depuis plus d'un an.
- En effet, on peut être TDAH sans symptôme dys-, même si c'est ultra-rare et c'est une critique classique de la technique Koala qui n'a pas pris cela en ligne de compte dans leur étude clinique.
- D'après mes sources, Koala aide sur certaines composantes, mais a globalement une efficacité moindre qu'un protocole de Neurofeedback suivi par un praticien. A voir le ratio coût / simplicité / efficacité recherché.
- Le neurofeedback n'est pas tellement à propos du matériel (en très gros, c'est bêtement des électrodes qui captent un signal électrique, rien de bien compliqué). Se procurer un matériel de Neurofeedback est d'ailleurs très facile. C'est savoir interpréter la carte cérébrale, définir le bon protocole d'entrainement et appliquer ce protocole qui nécessite de la pratique et de l'expérience. Et comme je le disais à Aurore au dessus, une liste de praticiens n'existant pas, je ne peux malheureusement pas donner d'éléments sur ce point là, désolé.
Bon courage à vous,
Rémi
Merci pour vos réponses.
Bien cordialement,
Patrick